Association between Copper–Zinc Ratio in Hair and Neutrophil–Lymphocyte Ratio within the Context of a Normal White Blood Cell Count among Overweight or Obese Korean Individuals: A Pilot Study
Article information
Abstract
Background
Obesity is considered a state of enhanced oxidative stress as well as chronic and low-grade inflammation. The copper–zinc ratio in obese individuals has been reported to reflect systemic oxidative stress and inflammatory status. We investigated whether the neutrophil–lymphocyte ratio is related to the copper–zinc ratio in hair, within the context of a normal white blood cell count among overweight or obese Korean individuals.
Methods
We included 56 participants aged older than 20 years who voluntarily sought weight reduction treatment and met the inclusion criterion of body mass index of 23 kg/m2 or greater. Intra-abdominal visceral adipose tissue was measured by computed tomography imaging, while the copper and zinc levels were measured by hair mineral analysis. Using multiple linear regression analysis, we examined the associations between the neutrophil–lymphocyte ratio and the copper–zinc ratio.
Results
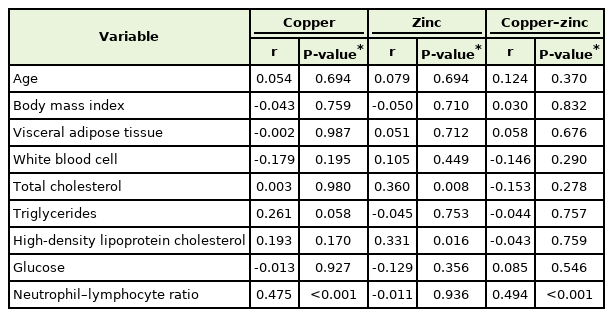
The mean age, body mass index, and visceral adipose tissue were 46.0±10.5 years, 29.0±4.1 kg/cm2, and 142.9±68.8 cm2, respectively. Pearson’s correlation analysis revealed the association of the neutrophil–lymphocyte ratio with copper level (r=0.475, P<0.001) and copper–zinc ratio (r=0.494, P<0.001). After adjusting for confounding variables, we found the neutrophil–lymphocyte ratio was significantly associated with the level of copper and the copper–zinc ratio in hair (regression coefficient: 0.055±0.015; P<0.001 and regression coefficient: 0.761±0.185; P<0.001, respectively).
Conclusion
A higher copper–zinc ratio in hair is positively and independently associated with the neutrophil–lymphocyte ratio. Thus, a high hair copper–zinc ratio could be a useful parameter for oxidative burden of individuals predisposed to obesity-related comorbidity.
INTRODUCTION
Obesity is considered to be a state of enhanced oxidative stress as well as chronic and low-grade inflammation [1]. The excessive accumulation of fat in adipose tissue recruits macrophages and leads to the increased production of proinflammatory cytokines and chemokines, such as interleukin 6 and tumor necrosis factor-α. In particular, visceral obesity is known to play a crucial role in the development of metabolic syndrome [2,3]. Thus, it is clinically important to evaluate the preclinical oxidative burden in obese patients.
White blood cell (WBC) count is one of the useful inflammatory biomarkers in clinical practice. A higher WBC count, even within a normal range, has been associated with atherosclerotic cardiovascular disease events and is also considered a marker of infection [4]. WBCs are composed of five different types of immune cells, and each subtype of WBCs exerts its own effects in different ways. Neutrophils and monocytes produce reactive oxygen species (ROS) and nitric oxide species (NOS), being chemically reactive molecules [5]. Unless ROS and NOS are properly neutralized by the antioxidant defense system or molecules, they can cause damage to cellular structures and possibly DNA. More recently, the neutrophil–lymphocyte ratio (NLR) has been reported to predict cardiovascular risk [6,7]. In this regard, NLR within the context of a normal WBC count has been proposed as a useful indicator to assess the oxidative stress status [8].
Elevated copper is known to be associated with oxidative stress, inflammation, and reduced immune function [9-11]. in contrast, zinc, an antagonist mineral for copper, has antioxidant and anti-inflammatory properties and is essential for maintaining the immune system. Thus, the ratio of copper to zinc has been reported to reflect systemic oxidative stress [12]. Some previous studies have shown that chronic inflammatory diseases are associated with increased copper–zinc ratio [13,14]. If the oxidative stress increases as the ratio of copper to zinc increases or vice versa, we would expect positive associations between the copper–zinc ratio and NLR with normal WBC count; thus, the copper–zinc ratio could be an additional surrogate marker for oxidative burden.
Hair mineral analysis is a noninvasive and useful test for assessing long-term mineral imbalance. Profiling the imbalance in hair minerals, such as an impaired ratio of mutually interactive minerals, might be a useful diagnostic tool for early-stage diagnosis in many diseases [15]. Thus, we investigated whether NLR is related to the copper–zinc ratio in hair within the context of a normal WBC count among overweight or obese Korean adults.
METHODS
1. Study Population
We retrospectively reviewed the medical records of 68 sequential adults who voluntarily sought weight reduction treatment at the Department of Family Medicine, Yongin Severance Hospital, Yongin-si, Republic of Korea, between April, 2011 and May, 2013. We recruited overweight or obese Korean adults who met the inclusion criteria of a body mass index (BMI) of 23 kg/m2 or more (the threshold value for generalized overweightness or obesity in Asia) and who underwent both hair mineral analysis and abdominal fat computed tomography (CT) imaging. Subjects meeting any the following criteria were excluded following a whole blood count analysis to rule out acute inflammatory disorders: WBC count of <4.0 or >10.0 (×103 cell/μL). Ultimately, 56 participants (11 men and 45 women) were included in final analysis.
Participants were asked about their medical history, and height and body weight were measured to the nearest 0.1 cm and 0.1 kg while the participant wore light indoor clothing and no shoes. BMI was calculated as the ratio of weight (kg) to the square of height (m2). Following an 8-hour overnight fast, blood samples were taken from an antecubital vein. WBC counts were quantified by an automated blood cell counter (ADVIA 120; Siemens Healthineers, Erlangen, Germany) within 1 hour of blood sampling. Fasting plasma glucose, total cholesterol, triglycerides, and high-density lipoprotein (HDL) cholesterol were measured using a Hitachi 7600-110 chemistry autoanalyzer (Hitachi, Tokyo, Japan). Hypertension was defined by systolic blood pressure ≥140 mm Hg, diastolic blood pressure ≥90 mm Hg, or current use of hypertension medication. Type 2 diabetes was defined by a fasting plasma glucose level ≥126 mg/dL or the current use of diabetes medication. Dyslipidemia was defined as triglyceride ≥150 mg/dL, low HDL cholesterol <50 mg/dL, or current use of dyslipidemia medications. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the institutional review board (IRB) of Yong-in Severance Hospital, Gyeonggi-do, Republic of Korea (IRB approval no., 9-2019-0012). The requirement for informed consent from individual patients was omitted because of the retrospective design of this study.
2. Hair Mineral Analysis
Participants were asked not to chemically process (e.g., by dying, perming, straightening, or frosting) their hair for at least 2 weeks before sample collection. Immediately before sample collection, we ensured that the sample was free of all gels, oils, and hair creams using a self-administered questionnaire. Hair samples (approximately 80 mg) were obtained from the proximal portion within 5 cm of the posterior vertex region of the scalp by cutting with stainless steel scissors and were stored in clean plastic bags. We measured the copper and zinc levels in each hair sample, using a microwave temperature-controlled digestion technique and a mass spectrometer (Sciex Elan 6100; PerkinElmer Corp., Waltham, MA, USA). The analytic process was regularly inspected and controlled by the Clinical Laboratory Division of the Department of Health and Human Services (Trace Elements Inc., Addison, TX, USA). The concentrations of copper and zinc are expressed as mg/100 g of hair with dry weight (mg%).
3. Abdominal Fat Computed Tomography
Abdominal fat CT is an accurate and convenient imaging test available to assess the visceral fat of an individual. We measured intra-abdominal (visceral adipose tissue, VAT) by fat measurement CT (Tomoscan 350; Philips, Amsterdam, the Netherlands). The VAT area was assessed at the L4 to L5 level with 3-mm slice thicknesses in the supine position, quantified by defining the intra-abdominal cavity at the internal side of the abdominal and oblique muscle walls surrounding the cavity and the posterior aspect of the vertebral body.
4. Statistical Analysis
Demographic and biochemical characteristics of the study population were calculated and expressed as means (±standard deviation) or percentages. Using Pearson’s correlation, we measured the strength of correlation between copper, zinc, and the copper–zinc ratio and metabolic indicators, including NLR. A multiple linear regression analysis was used to assess the independent contributions of copper, zinc, and the copper–zinc ratio to NLR after adjusting for confounding variables, including hypertension, type 2 diabetes, and dyslipidemia. All analyses were conducted using the SAS ver. 9.4 statistical software program (SAS Institute Inc, Cary, NC, USA). All statistical tests were two-sided, and statistical significance was determined at a P-value of less than 0.05.
RESULTS
Table 1 presents the general characteristics of the study population of 56 subjects (11 men and 45 women). The mean age, BMI, and VAT were 46.0±10.5 years, 29.0±4.1 kg/cm2, and 142.9±68.8 cm2, respectively. The mean WBC count and NLR were 6.6±1.5×103 cells/μL and 1.7±0.8, respectively. For hair mineral analysis, the mean copper level was 4.8±7.2 mg%, the mean zinc level was 18.4±10.1 mg% and the copper–zinc ratio was 0.3±0.6. Overall, hypertension medication was prevalent in 19.6% of the study population, while the proportions of diabetes medication and dyslipidemia medication use were 14.3% and 21.4%, respectively.
Table 2 shows the correlation analysis between copper, zinc, copper–zinc ratio, and metabolic indicators. We found that NLR was positively correlated with the copper level (r=0.475, P<0.001) and the copper–zinc ratio (r=0.494, P<0.001). However, such trends were not significant in the correlation of NLR with zinc level (r=-0.011, P=0.936). In addition, the level of copper was marginally significant when paired with the level of triglycerides (P=0.058), while the level of zinc was inversely correlated with the HDL cholesterol level (P=0.016).
Table 3 shows the independent contributions to NLR of copper, zinc, and the copper–zinc ratio in hair. In multiple regression model 2, after adjusting for age, sex, and BMI, the β for NLR was 0.057±0.014 (P<0.001), 0.002±0.012 (P=0.897), and 0.757±0.176 (P<0.001), in the case of copper, zinc, and the copper–zinc ratio, respectively. We also assessed the association between hair minerals and NLR after additional adjustment for VAT, hypertension, type 2 diabetes, and dyslipidemia. The associations were similar in model 3: β=0.055±0.015, P<0.001 for copper; β=-0.004±0.012, P=0.765 for zinc; and β=0.761±0.185, P<0.001 for the copper–zinc ratio.
DISCUSSION
In this cross-sectional study on overweight or obese patients, we observed a positive association between NLR and both the level of copper and the copper–zinc ratio in hair. Furthermore, these were found to be independent factors after additional adjustment for VAT, hypertension, type 2 diabetes, and dyslipidemia.
In previous studies, obesity has been linked to systemic inflammation and has been associated with various inflammatory markers, while NLR is an important predictor for evaluating chronic inflammation [15-19]. In particular, NLR may be a significant indicator when the pathophysiology involves inflammation of blood vessels, such as cardiovascular disease [8]. In the relationship between NLR and obesity, studies have shown that other markers, such as C-reactive protein (CRP), are more closely related with obesity, while NLR tends to be elevated in obese patients [20-23].
Copper is an essential nutrient but may induce oxidative damage. Excessive levels of copper can cause increased oxidative damage to lipids, proteins, and DNA. However, individuals with copper deficiency are less able to withstand oxidative stress. Thus, it is important to maintain the proper concentration of copper in the body [24-26].
Zinc functions to maintain a normal immune response, and chronic zinc deficiency increases the inflammatory response [27]. In many other chronic inflammatory conditions, zinc levels are low. Low levels of zinc were also found in obesity, one of the known chronic inflammatory conditions [28]. Oxidative stress and inflammatory response have shown improvement after zinc replacement [10,29,30]. In our study, however, participants presented with normal zinc levels in the hair mineral analysis, and we found no significant correlation between the level of zinc and NLR.
Zinc reduces copper toxicity by promoting the synthesis of metallothionein, which absorbs copper in the liver, intestine, and kidneys and removes copper from its binding site (which promotes radical formation) [24]. The copper–zinc ratio has been positively related to systemic oxidative stress status: copper has a pro-oxidant property, while zinc has an antioxidant property. Thus, the imbalance of copper and zinc may cause oxidative stress [12]. An increased NLR ratio as an oxidative stress burden has been reported to be independent of common inflammatory markers such as CRP [31]. A decrease in lymphocyte count is correlated with an increase in hypothalamic-pituitary-adrenal axis hyperactivity, and increases in neutrophil count may lead to enhanced oxidative stress through enzymatic changes such as myeloperoxidase activation [32]. In the present study we found that the copper–zinc ratio was significantly associated with NLR but not correlated with metabolic alterations, except for a weak relationship with respect to some parameters. In this regard, the increased copper–zinc ratio may represent increased oxidative burn within the context of a normal WBC count.
Our study has a few limitations that should be mentioned. First, as a cross-sectional study, the exact cause-and-effect relationship between the hair copper–zinc ratio and NLR remains unclear. Prospective longitudinal studies are needed to verify the role of high copper–zinc ratio in hair in the development of obesity-related, chronic inflammation. Second, since all study subjects were volunteers who had visited a single clinic for weight reduction, the way in which the study population was enrolled may have introduced selection bias and prevented us from obtaining an accurately representative sample of the general Korean population. Third, we did not take into consideration the effect of dietary pattern and the use of supplements as well as the hormonal conditions in women that could influence hair mineral levels and NLR ratio. Furthermore, although we excluded subjects with abnormal WBC counts, other inflammatory conditions cannot be completely excluded, and other inflammatory markers (such as CRP) were not measured at the beginning of this study. Despite these limitations, we argue that our effort to elucidate the role of copper–zinc interactions in the preclinical inflammatory state is a distinguishing feature of this study.
In conclusion, we found that a higher copper–zinc ratio in hair within the context of a normal WBC count is positive and independently associated with NLR. Accordingly, we propose the validity of deploying a high hair copper–zinc ratio as a putative oxidative burden among overweight or obese Korean participants, providing an initiative for further nutritional health management of individuals predisposed with obesity-related comorbidity. Further larger-scale studies are warranted to validate these associations.
Notes
CONFLICT OF INTEREST
No potential conflict of interest relevant to this article was reported.