|
 |
- Search
| Korean J Fam Med > Volume 33(6); 2012 > Article |
Abstract
Background
Combined therapy with alendronate and calcitriol may have additive effects on bone density. An observational study was performed to evaluate the efficacy and safety of Maxmarvil, a combinative agent of alendronate (5 mg) and calcitriol (0.5 ┬Ąg), and to identify factors associated with efficacy.
Methods
A total of 568 postmenopausal women with osteoporosis were enrolled by family physicians in 12 hospitals. The study subjects took Maxmarvil daily for 12 months. Questionnaires about baseline characteristics, socioeconomic status, and daily calcium intake were completed at the first visit. Adverse events were recorded every 3 months and bone mineral density (BMD) in the lumbar spine was measured using dual-energy X-ray absorptiometry at baseline and after 12 months. We evaluated the efficacy and safety of Maxmarvil, and the factors related to BMD improvement.
Results
A total of 370 patients were included in final analysis. The median BMD was 0.81 ┬▒ 0.12 g/cm2 at pre-treatment and 0.84 ┬▒ 0.13 g/cm2 after one year. The average BMD improvement was 3.4% ┬▒ 6.4% (P < 0.05), and 167 (45.1%) patients showed improvement. Factors associated with improved BMD were continuation of treatment (odds ratio [OR], 2.41; 95% confidence interval [CI], 1.15 to 5.07) and good compliance (OR, 2.54; 95% CI, 1.29 to 5.00). Adverse events were reported by 35 of the 568 patients, with the most common being abdominal pain and dyspepsia.
Osteoporosis is a common disorder characterized by reduced mineral density and bone mass, microarchitectural deterioration and increased skeletal fragility, and it results in an increased risk of fracture.1) Osteoporosis occurs in both males and females but is particularly common among women following menopause when bone turnover increases and the rate of bone resorption exceeds that of bone formation. Corresponding with an aging society, the public health impact of osteoporosis is enormous, affecting 200 million individuals worldwide. The reported prevalence of osteoporosis in Caucasian women older than 50 years of age varies from 7.9% to 22.6%.2,3) In a community-based cohort study consisting of 1,547 men and 1991 women in Korea, the crude prevalence of osteoporosis in subjects aged 40 to 79 years was 13.1% for men and 24.3% for women based on World Health Organization (WHO) criteria.4)
The goal of osteoporosis treatment is to reduce the fracture risk by preventing further loss of bone mineral density (BMD). Bisphosphonate inhibits bone resorption by reducing osteoclast number and function, and alendronate is one of the most potent and widely used bisphosphonates. In many European countries and in the United States, alendronate is being used clinically for the treatment of osteoporosis because of its established effects on bone density and fracture prevention.5,6) The recommended dose of alendronate is generally 10 mg daily or 70 mg weekly. Continuous treatment with 10 mg of alendronate daily for 10 years is associated with sustained therapeutic effects on bone density and remodeling.7) Studies in Japan found that 5 mg/d of alendronate increased BMD in Asian postmenopausal women with osteoporosis to a level similar to that achieved with 10 mg/d in Caucasian women.8,9)
Vitamin D enhances intestinal absorption of calcium and thereby affects bone mineralization. Low levels of vitamin D are associated with negative calcium balance, and a compensatory rise in parathyroid hormone, which results in excessive bone resorption. It has been estimated that 1 billion people worldwide could be classified as being vitamin D deficient or insufficient.10) Postmenopausal Korean women are at an increased risk of low vitamin D levels due to lack of exposure to sunshine, genetic traits, air pollution, and dietary habits.11) Several studies have demonstrated the positive effects of combining the use of active vitamin D (i.e., calcitriol) with antiresorptive agents such as calcitonin and bisphosphonates.12,13) Those results have led to the development of drugs such as Maxmarvil, which contains a combination of 5 mg alendronate and 0.5 ┬Ąg calcitriol.
The present study evaluated the efficacy and safety of Maxmarvil in Korean postmenopausal women with osteoporosis. We examined the effect of Maxmarvil on BMD, and identified factors associated with improved BMD.
The study recruited postmenopausal women who visited family physicians in 12 hospitals for osteoporosis or osteopenia treatment between September 2007 and August 2010. This study was approved by the institutional review board of the Asan Medical Center (2007-0400). Patients were excluded if they had a history of Maxmarvil treatment, a history of serious medical problems, or secondary osteoporosis related to systemic glucocorticoid therapy. The study comprised a safety analysis and an efficacy analysis. The safety analysis involved the initially enrolled 568 patients who had at least one follow-up visit after taking Maxmarvil. In the efficacy analysis, 198 patients were excluded because they were lost at follow-up (154 patients), there were missing values (21 patients), they refused to take medication (7 patients) or they discontinued treatment due to adverse events (16 patients). Therefore, 370 of the original 568 patients were involved in the efficacy analysis (Figure 1).
Demographic characteristics including marital, educational, and income status were assessed using a self-questionnaire at the first visit. Information regarding known health risks such as smoking, heavy drinking, exercise habits, and current medication use were also obtained. Patients were classified as current smoker, ex-smoker, and never a smoker, and as non-drinker or drinker. Exercise habits were categorized as less than once per week, one to two times per week, and three or more times per week.
A simple semi-quantitative food frequency questionnaire (SQFFQ) was used to assess the calcium intake of the patients. The questionnaire included questions regarding the dietary intake of 30 food items that contain calcium. Eight categories were used to determine intake frequency: hardly ever, once a month, two or three times a month, two or three times a week, four to six times a week, daily and over twice daily. Daily calcium intake and calcium ingestion rate based on food groups were calculated using the questionnaire data. The questionnaire was found to have validity when compared to previous semi-quantitative calcium intake frequency questionnaires (Pearson correlation coefficient, 0.50; Spearman correlation coefficient, 0.49). The prediction equation for estimating daily calcium intake from the SQFFQ was (daily calcium intake) = 14.928 ├Ś (calcium intake from SQFFQ)0.527.14)
Study subjects were given Maxmarvil (5 mg alendronate and 0.5 ┬Ąg calcitriol) daily for 1 year, after which they were evaluated. Patients were directed to take Maxmarvil in the early morning before breakfast with sufficient water, and were instructed to then remain upright for at least 30 minutes. Adverse events associated with the drugs were assessed at 3, 6, 9, and 12 months.
Lumbar spine BMD was measured using dual energy X-ray absorptiometry (DEXA). The BMD of the lumbar spine (L2-L4) was measured at commencement and after 12 months of treatment. A statistically significant increase in BMD was defined as a change exceeding the least significant change (LSC).
Calculation of the LSC requires consideration of the confidence interval. Specifically, if the LSC is determined at 95% confidence, the precision error (PE) is multiplied by 2.77 (LSC = 2.77 ├Ś PE).15) The PEs for each hospital's DEXA ranged from 0.7% to 1.5%. Therefore, we obtained the LSC from the median precision errors of each hospital's DEXA, and BMD changes that exceeded the LSC were considered to be significant. Using WHO criteria, osteoporosis was defined as a bone mineral density 2.5 standard deviations or more below the mean peak bone mass measured using DEXA.
The average BMD differences before and after treatment were compared using paired t-tests. Chi-square and Fisher's exact tests were used in univariate analyses to identify factors associated with BMD improvement. Logistic regression was used for multivariate analyses. The level of statistical significance was set at 0.05, and IBM SPSS ver. 19.0 (IBM Co., Armonk, NY, USA) was used for all statistical analyses. Data are expressed as mean, standard deviation, and odds ratio.
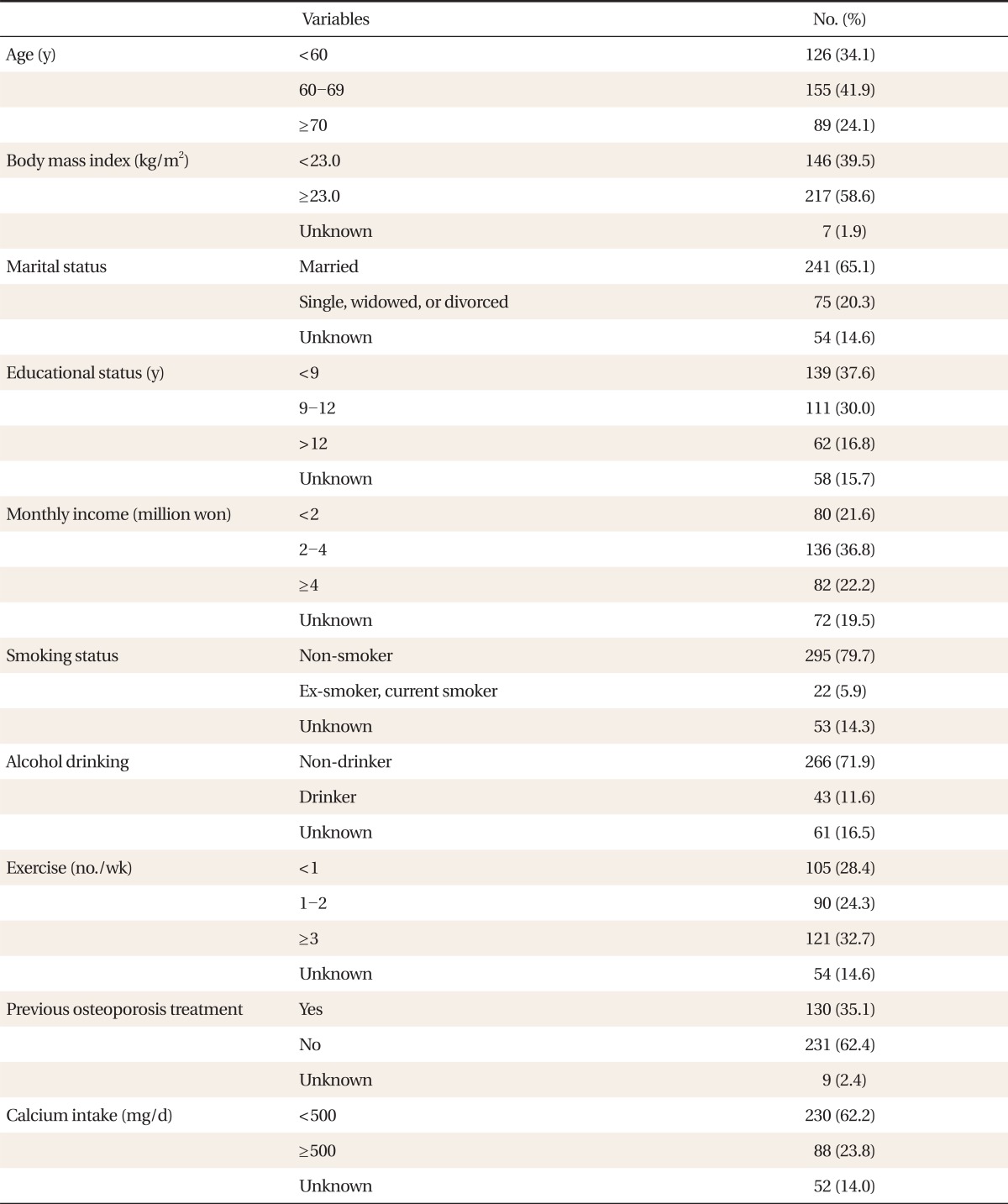
The average patient age was 63.4 ┬▒ 8.9 years and average body mass index (BMI) was 23.6 ┬▒ 3.1 kg/m2. Most patients had never smoked and were non-drinkers. In terms of physical activity, 28.4% partook less than once per week and 32.7% partook 3 or more times per week. Nearly one third (35.1%) had previously been treated for osteoporosis. Based on the SQFFQ responses, no patients had a daily calcium intake Ōēź 1,500 mg, which is the recommended dietary allowance (RDA) for women. There were 230 (62.2%) patients who had a daily calcium intake of < 500 mg (Table 1).
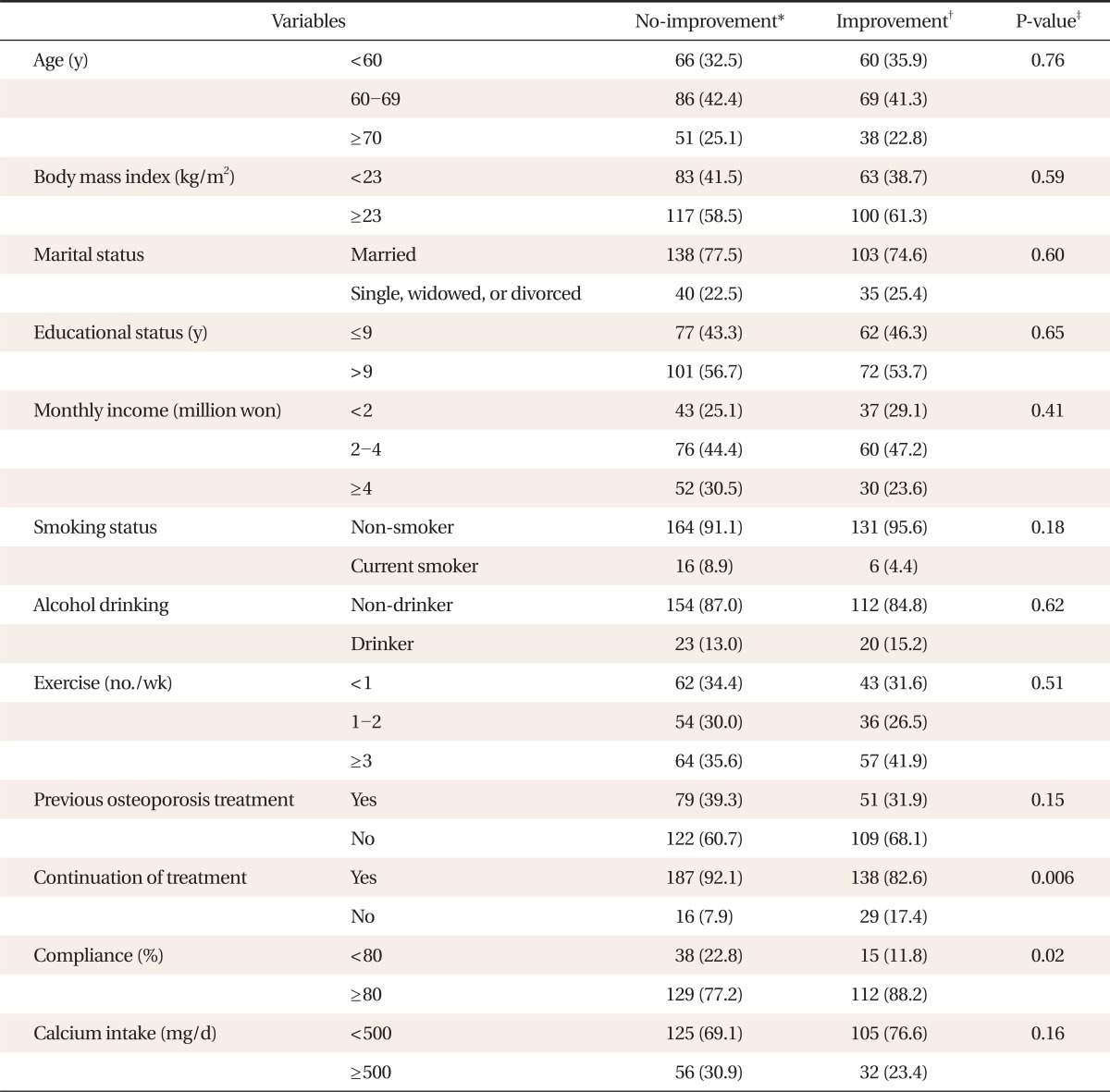
The pre-treatment average lumbar BMD was 0.81 ┬▒ 0.12 g/cm2, and the post-treatment average was 0.84 ┬▒ 0.13 g/cm2. The average improvement in lumbar BMD was 3.4% ┬▒ 6.4% (P < 0.001) (Figure 2). Of the 370 subjects who were included in the efficacy analyses, 167 (45.1%) showed improvement in lumbar BMD. Three hundred and twenty-five 325 (87.7%) patients continued the treatment after 1 year and 65.1% showed good compliance (i.e., >80% compliance) (Table 2).
Univariate analysis showed that continuation of treatment and good compliance (i.e., >80% compliance) were associated with BMD improvement (P < 0.05) (Table 3). Age, BMI, marital status, monthly income, educational status, smoking, alcohol, exercise, and calcium intake showed no significant association.
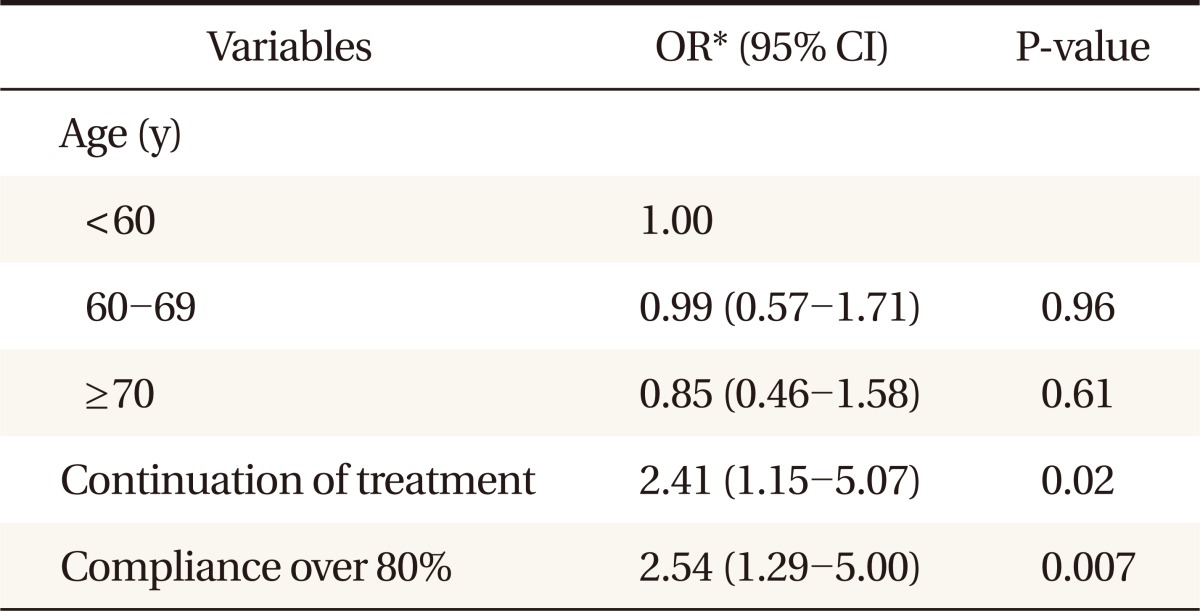
Multiple logistic regression analysis was performed to evaluate the independent factors associated with improved bone mineral density. The factors which were associated with bone density in univariate analysis and age were included in the analysis. Thus, multivariate logistic regression was performed including age, continuation of treatment, and compliance over 80%. According to the analysis, treatment continuation and good compliance (>80%) were associated with improvement of BMD (Table 4).
Safety analyses involved 568 patients. A total of 35 adverse events were reported in 32 subjects (5.6%). The most common adverse event was abdominal pain, which occurred in 13 patients (2.2%), followed by dyspepsia (1.7%) and nausea (0.5%). In addition, there was one case each of dizziness, weakness, pruritus, constipation, and edema (Table 5). Sixteen patients (2.8%) discontinued treatment as a direct result of adverse events. There were no serious adverse events leading to cessation of drug treatment.
The present study found that daily treatment with Maxmarvil for 12 months resulted in an increase in lumbar BMD. Considering the LSC for each hospital, we found that 167 subjects (45.1%) showed BMD improvement. Factors associated with BMD improvement were good compliance and continuation of treatment.
In the current study, the average improvement in lumbar BMD was 3.4% ┬▒ 6.4%. Another study conducted in Korea also using Maxmarvil reported a BMD increase of 2.42% ┬▒ 0.5% after 24 weeks of treatment.16) It is interesting that that amount of improvement was somewhat similar given that the treatment period was about half the duration of that in the present study. In a randomized open study including 226 postmenopausal women with osteoporosis, 10 mg alendronate plus 0.5 ┬Ąg alfacalcidol for 12 months was found to increase BMD by 4.3%.
The generally recommended dosage of alendronate is 10 mg daily or 70 mg weekly. However, previous studies conducted in Japan showed that 5 mg/d of aldendronate for 3 years was also effective in increasing BMD and reduced the risk of vertebral fractures.8) In addition, another study reported that 2.5 mg/d of alendronate had a positive effect on BMD, and that the effect was not significantly different from a 10 mg/d dosage.17) Compared with Caucasians, Asians have smaller body size and a lower dietary intake of calcium, therefore the recommended drug dosage established for Caucasians may not be appropriate for Asians.17)
Vitamin D deficiency is now a worldwide problem even in developed countries. In Korea, the prevalence of 25(OH) D levels less than 50 nmol/L during the wintertime is reported to be 90.1%.18) Furthermore, calcium intake for most Koreans is below the RDA. In the present study, no patients had a daily calcium intake >1,500 mg, which is the RDA for postmenopausal women. The superiority of calcitriol compared to native vitamin D in preventing bone loss and fractures has been established. In addition, combined treatment with alendronate and vitamin D analogs seems to be more effective, as compared to the respective monotherapies.19)
In the present study, multivariate analysis, which adjusted for age and other factors, found that continuation of treatment and good compliance were associated with BMD improvement. Drug compliance and adherence are important factors in successful treatment. In a previous study of postmarketing surveillance for the efficacy and safety of residronate, good compliance was associated with BMD improvement.20) And studies assessing the consequences of poor compliance and persistence in patients who did not follow their dosing regimens demonstrated lower BMD gains and increased risk of fracture.21,22) Dosing frequency is one of the factors contributing to poor adherence, and despite the daily regimen of Maxmarvil, the compliance and persistence in our study was high (65% and 87.7%, respectively). This may reflect the use of a combined drug, as against having to take bisphosphonate and calcitriol separately.
The present study used the simple SQFFQ to obtain calcium intake information. Using 500 mg/d of calcium as a referent, patients with an intake of Ōēź500 mg/d had an odds ratio for BMD improvement of 0.68 (95% confidence interval, 0.41 to 1.13). No association was observed between calcium intake and BMD improvement, either before or after adjustment. BMD improvement from calcium intake was expected to be relatively small because the efficacy of Maxmarvil was great. However, recall bias cannot be completely excluded because the measurement method used was a self-reporting questionnaire.
Drug safety is a critical issue. The overall adverse event rate in this study was 5.6%. Abdominal pain and dyspepsia were the most common adverse events, and accounted for more than half of the total events. The types of adverse events in this study did not differ from those reported in other bisphosphonate studies.23,24) The proportion of patients who experienced adverse events that led to discontinuation was 2.8% (16 patients). Therefore, Maxmarvil was found to be well tolerated with no serious adverse events and showed a low dropout rate during the study period.
The significance of this study is that it was a multicenter study conducted at 12 medical centers targeting people from various regions, which differentiates it from other studies. In addition, unlike other studies, this was an outpatient-oriented survey in a primary care setting, so the findings can be broadly applied with greater confidence. The study showed Maxmarvil had long-term efficacy up to 1 year, and it was possible to assess an accurate drug response due to the high drug compliance. Since all the centers measured BMD with DEXA, a standardized efficacy assessment was feasible.
The present study had several limitations. First of all, as our study analysis included the subjects who followed the study protocol completely, the efficacy of Maxmarvil could be exaggerated. In addition, this study did not include a placebo-controlled group. We could only specify the result by absolute changes in BMD before and after treatment. Therefore, we could not ascertain the direct relationship between Maxmarvil efficacy and fracture reduction. Although opinions differ as to whether fracture risk reduction is attributable to BMD improvement, major clinical trials have demonstrated that alendronate treatment reduced vertebral and non-vertebral fractures by 48% and 47%, respectively.25,26) Moreover, the relationship between BMD increase and fracture risk reduction is not proportional; a small BMD improvement has been found to cause a remarkable reduction in fracture risk.27,28) Although our study did not include a control group, by considering the absolute changes in BMD after treatment, we can assume that it would have positive effects on fracture reduction.
In conclusion, this survey of postmenopausal women with osteoporosis in a primary care setting found that Maxmarvil was efficacious, safe, and well tolerated.
ACKNOWLEDGMENTS
This study is part of a reexamination study of Maxmarvil, which is supported by a grant from YuYu Pharma, Inc.
References
1. Kanis JA, Melton LJ 3rd, Christiansen C, Johnston CC, Khaltaev N. The diagnosis of osteoporosis. J Bone Miner Res 1994;9:1137-1141. PMID: 7976495.


2. Emaus N, Omsland TK, Ahmed LA, Grimnes G, Sneve M, Berntsen GK. Bone mineral density at the hip in Norwegian women and men--prevalence of osteoporosis depends on chosen references: the Tromso Study. Eur J Epidemiol 2009;24:321-328. PMID: 19296062.


3. Tenenhouse A, Joseph L, Kreiger N, Poliquin S, Murray TM, Blondeau L, et al. Estimation of the prevalence of low bone density in Canadian women and men using a populationspecific DXA reference standard: the Canadian Multicentre Osteoporosis Study (CaMos). Osteoporos Int 2000;11:897-904. PMID: 11199195.


4. Shin CS, Choi HJ, Kim MJ, Kim JT, Yu SH, Koo BK, et al. Prevalence and risk factors of osteoporosis in Korea: a community-based cohort study with lumbar spine and hip bone mineral density. Bone 2010;47:378-387. PMID: 20362081.


5. Cranney A, Wells G, Willan A, Griffith L, Zytaruk N, Robinson V, et al. Meta-analyses of therapies for postmenopausal osteoporosis: II. Meta-analysis of alendronate for the treatment of postmenopausal women. Endocr Rev 2002;23:508-516. PMID: 12202465.


6. Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, et al. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group. Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. JAMA 1999;282:1344-1352. PMID: 10527181.


7. Bone HG, Hosking D, Devogelaer JP, Tucci JR, Emkey RD, Tonino RP, et al. Ten years' experience with alendronate for osteoporosis in postmenopausal women. N Engl J Med 2004;350:1189-1199. PMID: 15028823.


8. Kushida K, Shiraki M, Nakamura T, Kishimoto H, Morii H, Yamamoto K, et al. Alendronate reduced vertebral fracture risk in postmenopausal Japanese women with osteoporosis: a 3-year follow-up study. J Bone Miner Metab 2004;22:462-468. PMID: 15316867.


9. Iwamoto J, Takeda T, Sato Y, Uzawa M. Early changes in urinary cross-linked N-terminal telopeptides of type I collagen level correlate with 1-year response of lumbar bone mineral density to alendronate in postmenopausal Japanese women with osteoporosis. J Bone Miner Metab 2005;23:238-242. PMID: 15838627.


11. Park HM, Kim JG, Choi WH, Lim SK, Kim GS. The vitamin D nutritional Status of postmenopausal women in Korea. Korean J Bone Metab 2003;10:47-55.
12. Ushiroyama T, Ikeda A, Sakai M, Higashiyama T, Ueki M. Effects of the combined use of calcitonin and 1 alpha-hydroxycholecalciferol on vertebral bone loss and bone turnover in women with postmenopausal osteopenia and osteoporosis: a prospective study of long-term and continuous administration with low dose calcitonin. Maturitas 2001;40:229-238. PMID: 11731184.


13. Frediani B, Allegri A, Bisogno S, Marcolongo R. Effects of combined treatment with calcitriol plus alendronate on bone mass and bone turnover in postmenopausal osteoporosis: two years of continuous treatment. Clin Drug Investig 1998;15:235-244.

14. Lee JY, Ahn HS, Kim YS. Development and validation of a short food frequency questionnaire for estimating calcium intake. Korean J Health Promot Dis Prev 2001;1:8-17.
15. Lenchik L, Kiebzak GM, Blunt BA. International Society for Clinical Densitometry Position Development Panel and Scientific Advisory Committee. What is the role of serial bone mineral density measurements in patient management? J Clin Densitom 2002;5(Suppl):S29-S38. PMID: 12464709.


16. Rhee Y, Kang M, Min Y, Byun D, Chung Y, Ahn C, et al. Effects of a combined alendronate and calcitriol agent (Maxmarvil) on bone metabolism in Korean postmenopausal women: a multicenter, double-blind, randomized, placebo-controlled study. Osteoporos Int 2006;17:1801-1807. PMID: 17019520.


17. Shiraki M, Kushida K, Fukunaga M, Kishimoto H, Taga M, Nakamura T, et al. The Alendronate Phase III Osteoporosis Treatment Research Group. A double-masked multicenter comparative study between alendronate and alfacalcidol in Japanese patients with osteoporosis. Osteoporos Int 1999;10:183-192. PMID: 10525709.


18. So JS, Park HM. Relationship between parathyroid hormone, vitamin D and bone turnover markers in Korean postmenopausal women. Korean J Obstet Gynecol 2004;47:153-160.
19. Schacht E, Dukas L, Richy F. Combined therapies in osteoporosis: bisphosphonates and vitamin D-hormone analogs. J Musculoskelet Neuronal Interact 2007;7:174-184. PMID: 17627088.

20. Hung YJ, Kuo SW, Wang CH, Chang HY, Hsieh SH, Landen H. Postmarketing surveillance of acarbose treatment in Taiwanese patients with type 2 diabetes mellitus. Clin Drug Investig 2006;26:559-565.


21. Briesacher BA, Andrade SE, Yood RA, Kahler KH. Consequences of poor compliance with bisphosphonates. Bone 2007;41:882-887. PMID: 17707710.


22. Weycker D, Macarios D, Edelsberg J, Oster G. Compliance with osteoporosis drug therapy and risk of fracture. Osteoporos Int 2007;18:271-277. PMID: 17021945.


23. Ho AY, Kung AW. Efficacy and tolerability of alendronate once weekly in Asian postmenopausal osteoporotic women. Ann Pharmacother 2005;39:1428-1433. PMID: 16076919.


24. Adachi JD, Faraawi RY, O'Mahony MF, Nayar A, Massaad R, Evans JK, et al. Upper gastrointestinal tolerability of alendronate sodium monohydrate 10 mg once daily in postmenopausal women: a 12-week, randomized, double-blind, placebo-controlled, exploratory study. Clin Ther 2009;31:1747-1753. PMID: 19808133.


25. Liberman UA, Weiss SR, Broll J, Minne HW, Quan H, Bell NH, et al. The Alendronate Phase III Osteoporosis Treatment Study Group. Effect of oral alendronate on bone mineral density and the incidence of fractures in postmenopausal osteoporosis. N Engl J Med 1995;333:1437-1443. PMID: 7477143.


26. Pols HA, Felsenberg D, Hanley DA, Stepan J, Munoz-Torres M, Wilkin TJ, et al. Fosamax International Trial Study Group. Multinational, placebo-controlled, randomized trial of the effects of alendronate on bone density and fracture risk in postmenopausal women with low bone mass: results of the FOSIT study. Osteoporos Int 1999;9:461-468. PMID: 10550467.


27. Wasnich RD, Miller PD. Antifracture efficacy of antiresorptive agents are related to changes in bone density. J Clin Endocrinol Metab 2000;85:231-236. PMID: 10634392.


28. Watts NB, Cooper C, Lindsay R, Eastell R, Manhart MD, Barton IP, et al. Relationship between changes in bone mineral density and vertebral fracture risk associated with risedronate: greater increases in bone mineral density do not relate to greater decreases in fracture risk. J Clin Densitom 2004;7:255-261. PMID: 15319494.


Figure┬Ā2
Mean lumbar spine bone mineral density (BMD, g/cm2) after one year treatment with Maxmarvil (n = 370). Error bar indicates ┬▒ 1 SD. *P < 0.001 by paired t-test.