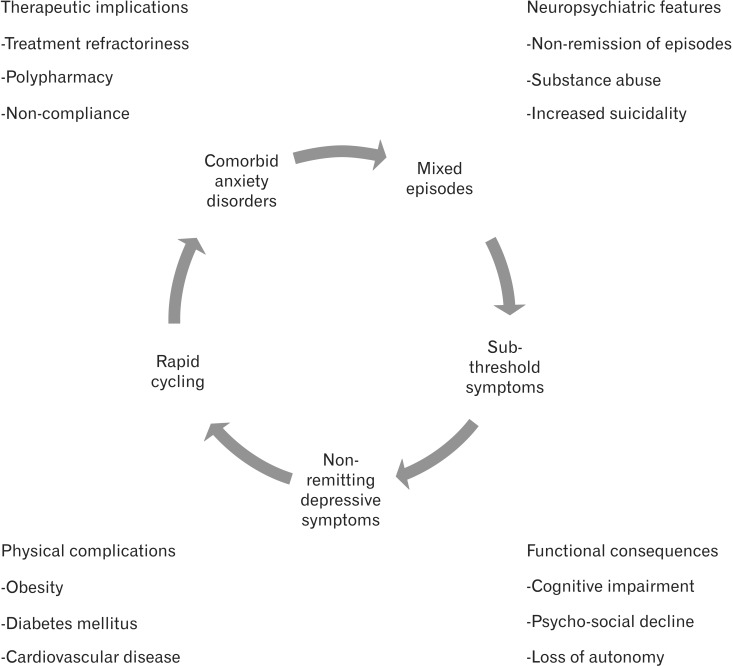
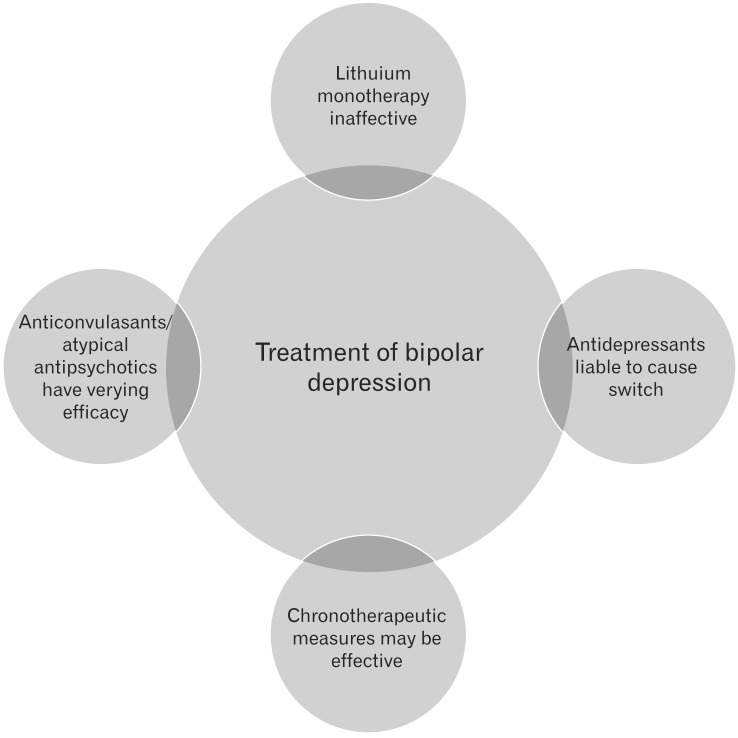
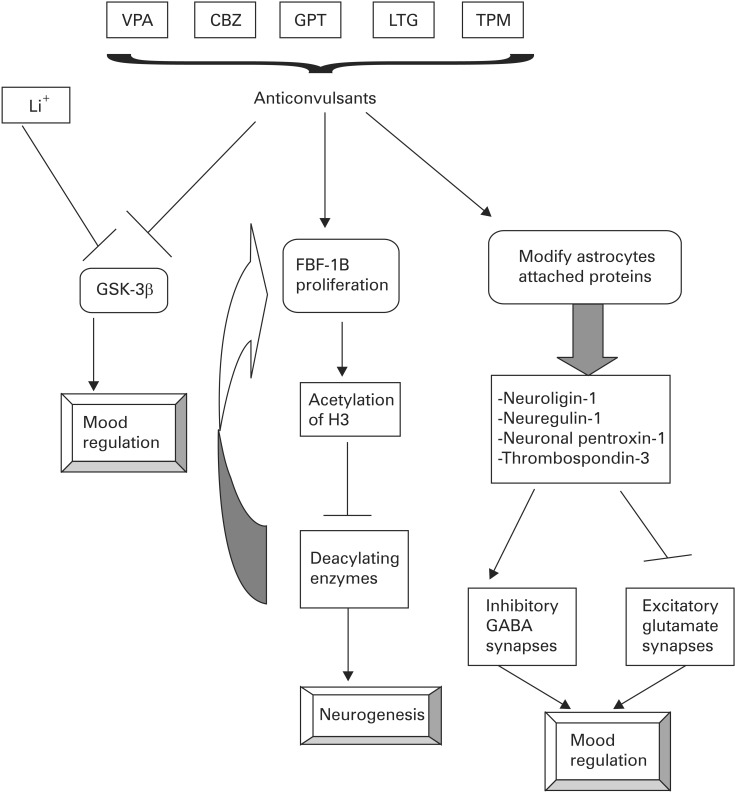
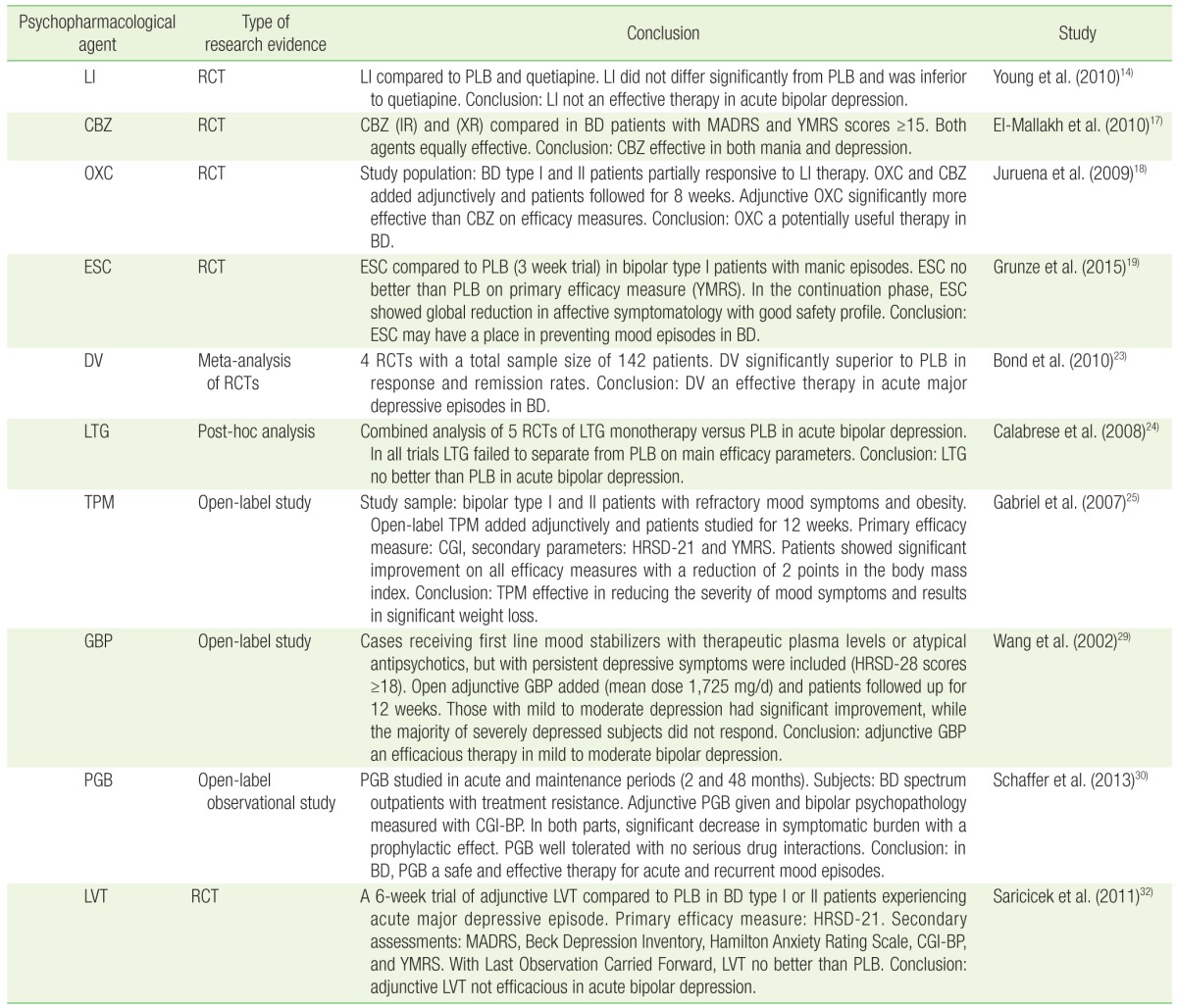
2. Ketter TA. Diagnostic features, prevalence, and impact of bipolar disorder. J Clin Psychiatry 2010;71:e14PMID:
20573324.


3. Nurnberger JI Jr, McInnis M, Reich W, Kastelic E, Wilcox HC, Glowinski A, et al. A high-risk study of bipolar disorder: childhood clinical phenotypes as precursors of major mood disorders. Arch Gen Psychiatry 2011;68:1012-1020. PMID:
21969459.



4. Krishnan KR. Psychiatric and medical comorbidities of bipolar disorder. Psychosom Med 2005;67:1-8. PMID:
15673617.


5. Sienaert P, Lambrichts L, Dols A, De Fruyt J. Evidence-based treatment strategies for treatment-resistant bipolar depression: a systematic review. Bipolar Disord 2013;15:61-69. PMID:
23190379.


6. Tondo L, Baldessarini RJ, Vazquez G, Lepri B, Visioli C. Clinical responses to antidepressants among 1036 acutely depressed patients with bipolar or unipolar major affective disorders. Acta Psychiatr Scand 2013;127:355-364. PMID:
23121222.


7. Benedetti F, Barbini B, Fulgosi MC, Colombo C, Dallaspezia S, Pontiggia A, et al. Combined total sleep deprivation and light therapy in the treatment of drug-resistant bipolar depression: acute response and long-term remission rates. J Clin Psychiatry 2005;66:1535-1540. PMID:
16401154.


8. Muneer A. Bipolar disorder: role of inflammation and the development of disease biomarkers. Psychiatry Investig 2016;13:18-33.


9. Nery LR, Eltz NS, Martins L, Guerim LD, Pereira TC, Bogo MR, et al. Sustained behavioral effects of lithium exposure during early development in zebrafish: involvement of the Wnt-β-catenin signaling pathway. Prog Neuropsychopharmacol Biol Psychiatry 2014;55:101-108. PMID:
24813569.


10. Prickaerts J, Moechars D, Cryns K, Lenaerts I, van Craenendonck H, Goris I, et al. Transgenic mice overexpressing glycogen synthase kinase 3beta: a putative model of hyperactivity and mania. J Neurosci 2006;26:9022-9029. PMID:
16943560.



12. Boku S, Nakagawa S, Masuda T, Nishikawa H, Kato A, Takamura N, et al. Valproate recovers the inhibitory effect of dexamethasone on the proliferation of the adult dentate gyrus-derived neural precursor cells via GSK-3β and β-catenin pathway. Eur J Pharmacol 2014;723:425-430. PMID:
24211784.


13. Wang CC, Chen PS, Hsu CW, Wu SJ, Lin CT, Gean PW. Valproic acid mediates the synaptic excitatory/inhibitory balance through astrocytes--a preliminary study. Prog Neuropsychopharmacol Biol Psychiatry 2012;37:111-120. PMID:
22343008.


14. Young AH, McElroy SL, Bauer M, Philips N, Chang W, Olausson B, et al. A double-blind, placebo-controlled study of quetiapine and lithium monotherapy in adults in the acute phase of bipolar depression (EMBOLDEN I). J Clin Psychiatry 2010;71:150-162. PMID:
20122369.


15. Missio G, Moreno DH, Fernandes F, Bio DS, Soeiro-de-Souza MG, Rodrigues dos, et al. The ARIQUELI study: potentiation of quetiapine in bipolar I nonresponders with lithium versus aripiprazole. Trials 2013;14:190PMID:
23805994.



17. El-Mallakh RS, Salem MR, Chopra A, Mickus GJ, Penagaluri P, Movva R. A blinded, randomized comparison of immediate-release and extended-release carbamazepine capsules in manic and depressed bipolar subjects. Ann Clin Psychiatry 2010;22:3-8. PMID:
20196977.

18. Juruena MF, Ottoni GL, Machado-Vieira R, Carneiro RM, Weingarthner N, Marquardt AR, et al. Bipolar I and II disorder residual symptoms: oxcarbazepine and carbamazepine as add-on treatment to lithium in a double-blind, randomized trial. Prog Neuropsychopharmacol Biol Psychiatry 2009;33:94-99. PMID:
19007842.


19. Grunze H, Kotlik E, Costa R, Nunes T, Falcao A, Almeida L, et al. Assessment of the efficacy and safety of eslicarbazepine acetate in acute mania and prevention of recurrence: experience from multicentre, double-blind, randomised phase II clinical studies in patients with bipolar disorder I. J Affect Disord 2015;174:70-82. PMID:
25484179.


20. Dimitrakopoulos S, Konstantakopoulos G. Pharmacological agents under research for the maintenance treatment in bipolar disorder. Psychiatriki 2015;26:169-180. PMID:
26480221.

21. Ghaemi SN, Gilmer WS, Goldberg JF, Zablotsky B, Kemp DE, Kelley ME, et al. Divalproex in the treatment of acute bipolar depression: a preliminary double-blind, randomized, placebo-controlled pilot study. J Clin Psychiatry 2007;68:1840-1844. PMID:
18162014.


22. Muzina DJ, Gao K, Kemp DE, Khalife S, Ganocy SJ, Chan PK, et al. Acute efficacy of divalproex sodium versus placebo in mood stabilizernaive bipolar I or II depression: a double-blind, randomized, placebocontrolled trial. J Clin Psychiatry 2011;72:813-819. PMID:
20816041.


23. Bond DJ, Lam RW, Yatham LN. Divalproex sodium versus placebo in the treatment of acute bipolar depression: a systematic review and meta-analysis. J Affect Disord 2010;124:228-234. PMID:
20044142.


24. Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disord 2008;10:323-333. PMID:
18271912.


25. Gabriel A. Adjunctive topiramate treatment in refractory obese bipolar patients: a descriptive open label study. Eat Weight Disord 2007;12:48-53. PMID:
17384530.


26. Roy Chengappa K, Kupfer DJ, Parepally H, John V, Basu R, Buttenfield J, et al. A placebo-controlled, random-assignment, parallel-group pilot study of adjunctive topiramate for patients with schizoaffective disorder, bipolar type. Bipolar Disord 2007;9:609-617. PMID:
17845276.


27. Kukkar A, Bali A, Singh N, Jaggi AS. Implications and mechanism of action of gabapentin in neuropathic pain. Arch Pharm Res 2013;36:237-251. PMID:
23435945.


28. Rakofsky JJ, Dunlop BW. Treating nonspecific anxiety and anxiety disorders in patients with bipolar disorder: a review. J Clin Psychiatry 2011;72:81-90. PMID:
21208580.


29. Wang PW, Santosa C, Schumacher M, Winsberg ME, Strong C, Ketter TA. Gabapentin augmentation therapy in bipolar depression. Bipolar Disord 2002;4:296-301. PMID:
12479661.


30. Schaffer LC, Schaffer CB, Miller AR, Manley JL, Piekut JA, Nordahl TE. An open trial of pregabalin as an acute and maintenance adjunctive treatment for outpatients with treatment resistant bipolar disorder. J Affect Disord 2013;147:407-410. PMID:
23040739.


31. Lee CY, Chen CC, Liou HH. Levetiracetam inhibits glutamate transmission through presynaptic P/Q-type calcium channels on the granule cells of the dentate gyrus. Br J Pharmacol 2009;158:1753-1762. PMID:
19888964.



32. Saricicek A, Maloney K, Muralidharan A, Ruf B, Blumberg HP, Sanacora G, et al. Levetiracetam in the management of bipolar depression: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry 2011;72:744-750. PMID:
21034692.


33. Peterson SM, Pack TF, Caron MG. Receptor, ligand and transducer contributions to dopamine D2 receptor functional selectivity. PLoS One 2015;10:e0141637PMID:
26516769.



35. Park SM, Chen M, Schmerberg CM, Dulman RS, Rodriguiz RM, Caron MG, et al. Effects of β-arrestin-biased dopamine D2 receptor ligands on schizophrenia-like behavior in hypoglutamatergic mice. Neuropsychopharmacology 2016;41:704-715. PMID:
26129680.



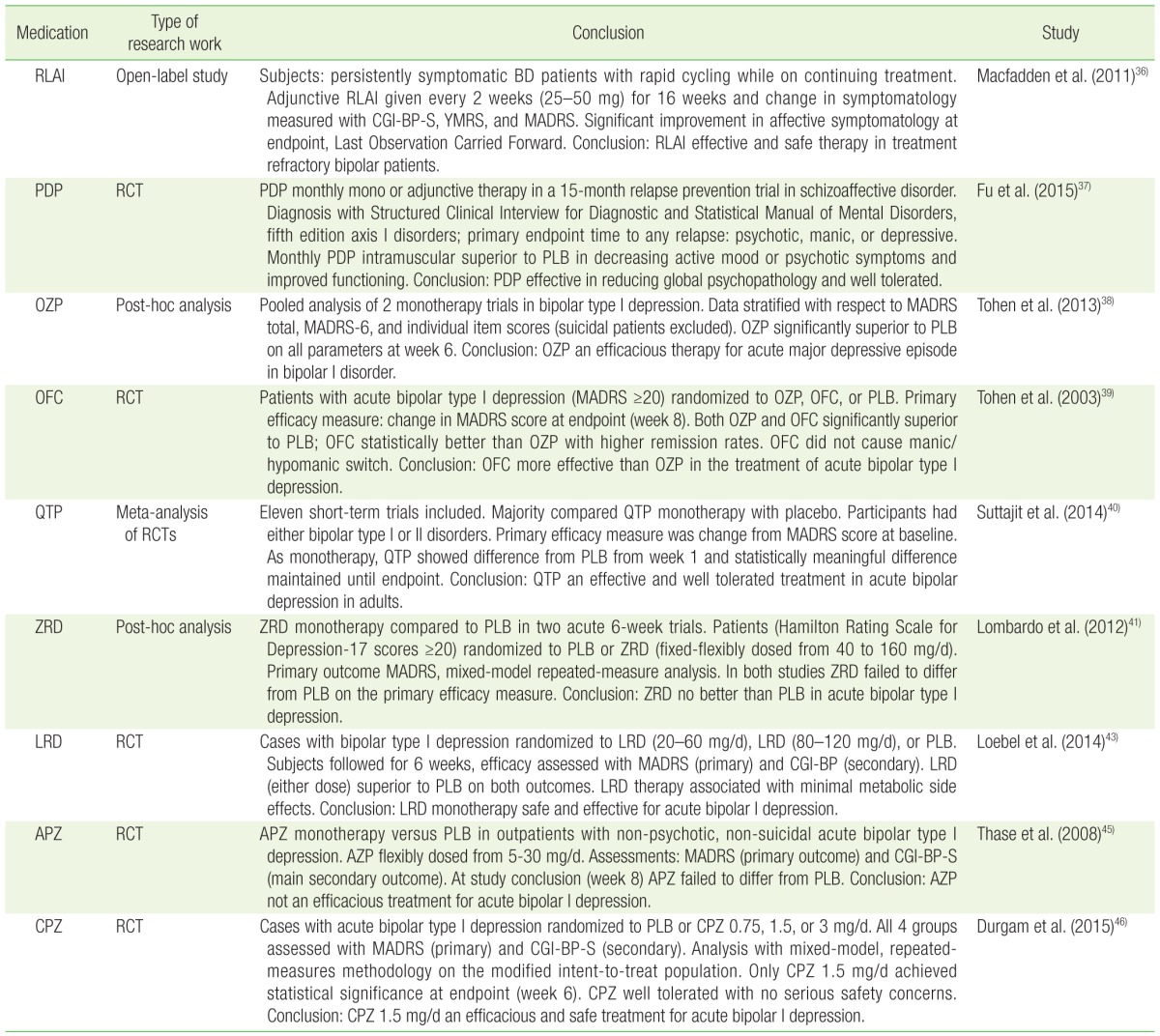
37. Fu DJ, Turkoz I, Simonson RB, Walling DP, Schooler NR, Lindenmayer JP, et al. Paliperidone palmitate once-monthly reduces risk of relapse of psychotic, depressive, and manic symptoms and maintains functioning in a double-blind, randomized study of schizoaffective disorder. J Clin Psychiatry 2015;76:253-262. PMID:
25562685.


38. Tohen M, Katagiri H, Fujikoshi S, Kanba S. Efficacy of olanzapine monotherapy in acute bipolar depression: a pooled analysis of controlled studies. J Affect Disord 2013;149:196-201. PMID:
23485111.


39. Tohen M, Vieta E, Calabrese J, Ketter TA, Sachs G, Bowden C, et al. Efficacy of olanzapine and olanzapine-fluoxetine combination in the treatment of bipolar I depression. Arch Gen Psychiatry 2003;60:1079-1088. PMID:
14609883.


41. Lombardo I, Sachs G, Kolluri S, Kremer C, Yang R. Two 6-week, randomized, double-blind, placebo-controlled studies of ziprasidone in outpatients with bipolar I depression: did baseline characteristics impact trial outcome? J Clin Psychopharmacol 2012;32:470-478. PMID:
22722504.


42. Sachs GS, Ice KS, Chappell PB, Schwartz JH, Gurtovaya O, Vanderburg DG, et al. Efficacy and safety of adjunctive oral ziprasidone for acute treatment of depression in patients with bipolar I disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry 2011;72:1413-1422. PMID:
21672493.


43. Loebel A, Cucchiaro J, Silva R, Kroger H, Hsu J, Sarma K, et al. Lurasidone monotherapy in the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry 2014;171:160-168. PMID:
24170180.


44. Loebel A, Cucchiaro J, Silva R, Kroger H, Sarma K, Xu J, et al. Lurasidone as adjunctive therapy with lithium or valproate for the treatment of bipolar I depression: a randomized, double-blind, placebo-controlled study. Am J Psychiatry 2014;171:169-177. PMID:
24170221.


45. Thase ME, Jonas A, Khan A, Bowden CL, Wu X, McQuade RD, et al. Aripiprazole monotherapy in nonpsychotic bipolar I depression: results of 2 randomized, placebo-controlled studies. J Clin Psychopharmacol 2008;28:13-20. PMID:
18204335.


46. Durgam S, Earley W, Lipschitz A, Guo H, Laszlovszky I, Nemeth G, et al. An 8-week randomized, double-blind, placebo-controlled evaluation of the safety and efficacy of cariprazine in patients with bipolar I depression. Am J Psychiatry 2016;173:271-281. PMID:
26541814.