Best Practice for Prolonged Fever in Primary Care Setting: Close Follow-Up or Empiric Antibiotic Therapy?
Article information
Abstract
The management of prolonged fever in low-socioeconomic-status areas by primary care providers such as general practitioners is challenging. Given the endemic nature of many infectious diseases, physicians typically start empirical antibiotic therapy following a limited diagnostic workup including serologic examinations. Herein, we report the case of a young male patient with prolonged fever and arthralgia initially diagnosed with and treated for brucellosis but with a confirmed diagnosis of systemic lupus erythematosus on follow-up. This unique case shows that close follow-up is the best practice for managing prolonged fever in cases with non-specific laboratory findings.
INTRODUCTION
Fever is one of the most common symptoms of a wide variety of diseases, including infectious, rheumatologic, and benign and malignant neoplastic diseases. General practitioners and family medicine physicians treat patients in primary care settings. To manage prolonged fever in the primary care setting, many physicians consider the most prevalent endemic infectious diseases and prescribe empirical antibiotic therapy based on serological and other preliminary laboratory study findings. This management strategy is very simple and may be helpful in many circumstances. However, empirical antibiotic therapy may miss some important non-infectious diseases, complicating the patient’s clinical course. Herein we present a case of systemic lupus erythematosus (SLE) that was initially diagnosed as and treated for brucellosis.
SLE, one of the most common rheumatologic disorders, has a wide variety of manifestations [1]. The classic clinical findings may not appear at the initial presentation but may evolve over several months [2]. In addition, the non-specific feature of multi-organ symptoms is another diagnostic challenge that could result in misdiagnosis [3,4].
Brucellosis is a bacterial zoonotic disease manifesting with some non-specific symptoms such as fluctuating fever, night sweats, and joint pain and discomfort. Given the lack of specific clinical signs, brucellosis can mimic other infectious and inflammatory diseases [3].
Some reports have detailed the co-occurrence of SLE and brucellosis [5]. It was also reported that SLE could be misdiagnosed as brucellosis [3]. Positive serology for Brucella may occur in diseases such as typhoid fever, malaria, and lymphoma [6]. However, similar reports on SLE are limited [3]. Furthermore, the positive Brucella serologic findings are common in endemic regions, which can interfere with the diagnosis of other diseases such as SLE [3].
Here we describe the case of a young man who was initially diagnosed with and treated for brucellosis. This case report reviews the potential clinical and laboratory challenges involved in diagnosing SLE in brucellosis-endemic regions.
CASE REPORT
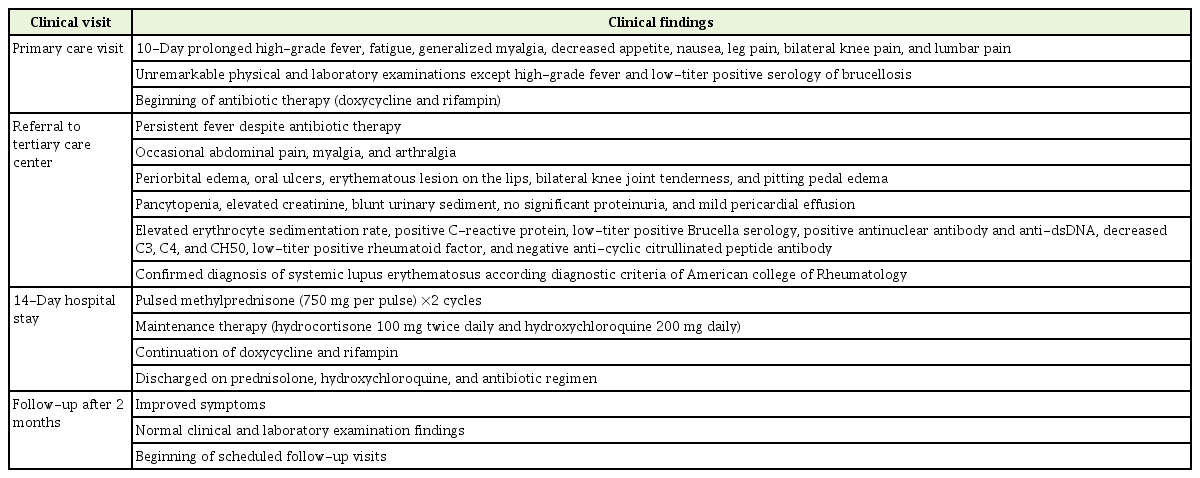
Our patient was an 18-year-old man from the Sistan-Balochestan province in southeastern Iran. He had a 10-day history of prolonged high-grade fever before admission. Other systemic symptoms were fatigue, generalized myalgia, decreased appetite, and nausea. Leg pain and bilateral arthralgia in the knee joints as well as lumbar pain was reported. His family history was unremarkable, and he had administered some antipyretics and analgesics.
The physical examination showed an ill patient with a body temperature >38°C. The head and neck examinations were negative for upper respiratory infection. The heart and lungs were normal. No significant lymphadenopathy or hepatosplenomegaly was found and no skin rashes were noted. The initial rheumatologic examinations were nonspecific.
The routine laboratory examinations were insignificant and the serological and microbiologic studies were non-diagnostic. However, the serology of Brucella was positive with a low titer including Wright test (1/40) and 2ME test (1/20).
Given the regional endemicity and the patient’s occupational history of sheep bearing, the patient was treated for brucellosis. The antibiotic regimen was a combination of doxycycline and rifampin.
Despite the antibiotic therapy, the patient’s persisted. Progressive periorbital and peripheral edema as well as occasional abdominal pain appeared. The patient had no mucocutaneous complaints except some painless oral ulcers. Previous myalgia and arthralgia have been pronounced. The patient was referred to our tertiary care center for further testing.
The physical examinations revealed fever, bilateral periorbital edema, some oral ulcers in the palatal mucosa, erythematous lesions on the lips, and bilateral pitting pedal edema. The systemic findings were otherwise normal. Detailed rheumatologic examinations showed no significant findings except bilateral tenderness of the knee joints.
The complete blood counts and peripheral smears revealed pancytopenia. Increased creatinine was found, but the urinary sediment was blunt and no significant 24-hour proteinuria was detected. The bone marrow aspiration was reactive. The hand X-ray showed no evidence of articular erosion. Echocardiography revealed a mild pericardial effusion without any evidence of endocarditis. Abdominopelvic ultrasound findings were normal.
A repeat serological test for Brucella was positive with a low titer. The evaluations were negative for Salmonellosis and Malaria. No evidence of human immunodeficiency virus (HIV) infection or viral hepatitis was found. Given the clinical findings of pancytopenia, oral ulcers, and pericardial effusion, serological tests for SLE were performed. The results showed an elevated erythrocyte sedimentation rate; positivity for C-reactive protein; positivity for antinuclear antibody and anti-dsDNA; decreased C3, C4, and CH50 levels; low-titer–positive rheumatoid factor; and negativity for anti-cyclic citrullinated peptide antibody. The diagnosis of SLE was confirmed according to the diagnostic criteria of the American College of Rheumatology.
The patient received two 750-mg cycles of pulsed methylprednisolone within the 3 subsequent days. Hydrocortisone 100 mg twice daily and hydroxychloroquine 200 mg daily were considered for maintenance therapy. Treatment with doxycycline and rifampin was continued in conjunction with the pulsed and maintenance therapy.
The patient’s symptoms improved following the administration of glucocorticoids. The hydrocortisone was substituted for by oral prednisolone 20 mg twice daily. The patient was discharged following 14 days of hospitalization on the prior prednisolone, hydroxychloroquine, and antibiotic regimen. At a follow-up visit 2 months after discharge, no evidence of fever, myalgia, arthralgia, oral ulcers, or periorbital and pedal edema was seen. The laboratory examination revealed normal complete blood counts, creatinine and electrolytes, negative urinalysis, and no 24-hour proteinuria. The patient’s stepwise clinical scenario is summarized in Table 1.
DISCUSSION
Making the diagnosis of SLE in regions with a high endemicity of brucellosis may be difficult due to diagnostic challenges [7]. Non-specific initial presentations such as prolonged fever, myalgia, and arthralgia are not helpful findings to differentiate SLE from brucellosis [7,8]. In addition, it may take several days or months before the classical pattern of joint involvement appear [9]. Characteristic clues such as malar and discoid rashes, photosensitivity, and oral ulcers could be absent at presentation [9,10]. Evidence of organ involvement such as raised creatinine, cytopenia, seizure, or psychosis may be found in close follow-up visits [9]. On the other hand, the patient’s socioeconomic context and occupational history can result in the misdiagnosis of brucellosis in endemic regions.
Laboratory pitfalls are another challenge in the diagnosis of SLE in brucellosis-endemic regions. Considering the predisposition to exposure in endemic regions, a low-titer of serologic Brucella on the Wright and 2ME tests may be found [7-10]. Higher serologic titers indicate brucellosis, but low or intermediate titers are not able to differentiate SLE from brucellosis [3].
The serology of brucellosis can reportedly be positive for other diseases such as typhoid fever, malaria, and lymphoma [6]. However, the false-positive serology of brucellosis in SLE patients has not been clearly explained in the literature [3]. In contrast to brucellosis, falsepositivity for antibodies of other infectious diseases such as those caused by cytomegalovirus and Epstein–Barr virus have been reported in SLE patients [11,12]. Similarly, Jian et al. [13] reported the case of an elderly woman with false-positive HIV serology who was diagnosed with SLE.
Given the clinical and laboratory pitfalls associated with diagnosing SLE in brucellosis-endemic regions, it can be concluded that close follow-up is the best practice. The equivocal serologic findings should be confirmed by definite measures such as microbiological cultures.
Notes
No potential conflict of interest relevant to this article was reported.