|
 |
- Search
| Korean J Fam Med > Volume 42(5); 2021 > Article |
|
Abstract
Background
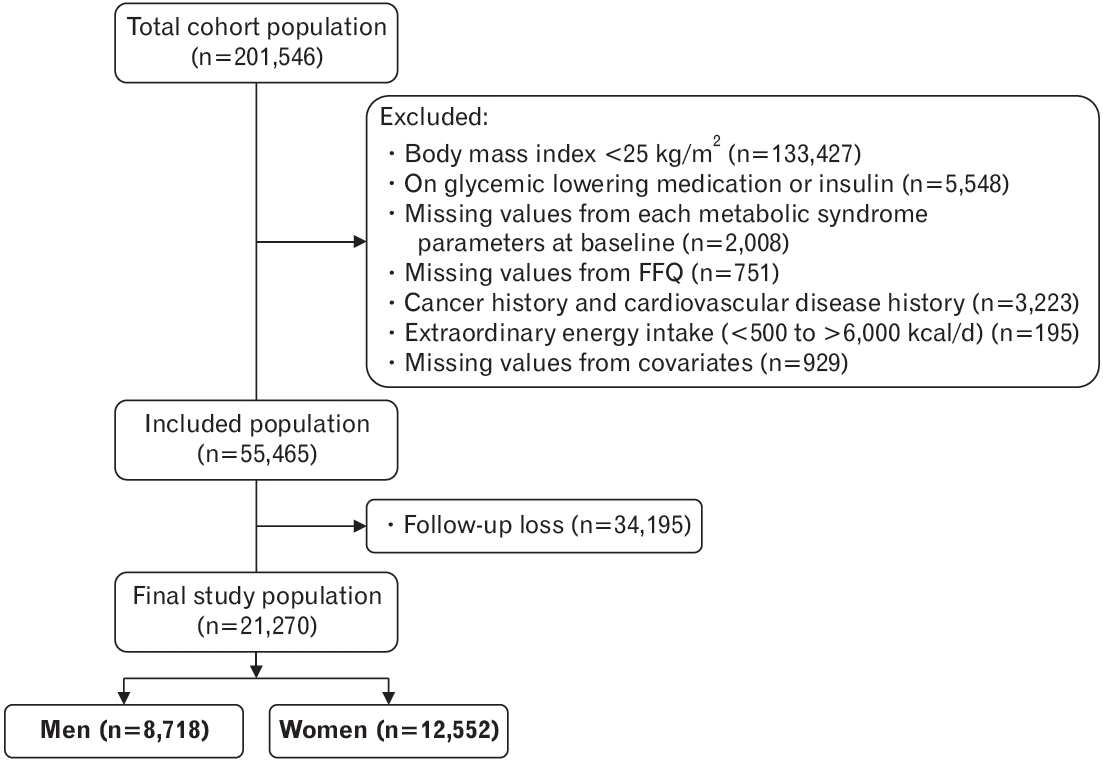
Methods
Results
Table 1.
| Characteristic | Men | Women | P-value |
|---|---|---|---|
| No. of participants | 8,718 | 12,552 | |
| Age (y) | 54.8±8.5 | 55.0±8.0 | <0.001 |
| Body mass index (kg/m2) | 27.0±1.7 | 27.2±2.0 | <0.001 |
| Present of hypertension | 2,521 (28.9) | 3,442 (27.4) | <0.01 |
| Present of dyslipidemia | 986 (11.3) | 1,363 (10.9) | <0.05 |
| Physical activity | <0.001 | ||
| Yes | 4,958 (56.9) | 5,786 (46.1) | |
| No | 3,760 (43.1) | 6,766 (53.9) | |
| Smoking | <0.001 | ||
| Never smoker | 2,485 (28.5) | 12,192 (97.1) | |
| Former smoker | 3,860 (44.3) | 134 (1.1) | |
| Current smoker | 2,373 (27.2) | 226 (1.8) | |
| Alcohol use | <0.001 | ||
| Non-drinker | 1,724 (19.8) | 8,686 (69.3) | |
| Former drinker | 628 (7.2) | 248 (2.0) | |
| Current drinker | 6,366 (73.0) | 3,608 (28.7) | |
| Family income (million won/mo) | <0.001 | ||
| <1 | 681 (9.2) | 1,806 (18.2) | |
| 1–2 | 1,310 (17.7) | 2,323 (23.4) | |
| 2–4 | 3,259 (44.1) | 3,951 (39.7) | |
| >4 | 2,134 (28.9) | 1,862 (18.7) | |
| Education year (y) | <0.001 | ||
| ≤6 | 1,944 (22.3) | 5,108 (40.7) | |
| >6–12 | 3,447 (39.6) | 5,742 (45.8) | |
| >12 | 3,323 (38.1) | 1,693 (13.5) | |
| Cardio-metabolic parameters | |||
| Waist circumference (cm) | 91.2±5.9 | 86.7±6.8 | <0.001 |
| Systolic blood pressure (mm Hg) | 128.9±14.4 | 125.8±15.3 | <0.001 |
| Diastolic blood pressure (mm Hg) | 81.3±9.7 | 78.1±9.8 | 0.599 |
| Triglycerides (mg/dL) | 173.7±111.0 | 135.8±82.1 | <0.001 |
| High-density lipoprotein-cholesterol (mg/dL) | 45.7±10.5 | 50.9±11.6 | <0.001 |
| Fasting blood glucose (mg/dL) | 98.4±17.7 | 94.2±15.8 | <0.001 |
| Total fruit intake (g/1,000 kcl)* | 78.4±72.0 | 112.8±96.0 | <0.001 |
| Total energy intake (kcal/d) | 1888.9±541.0 | 1696.3±539.1 | 0.713 |
| Macronutrient intake† | |||
| Carbohydrate (g/1,000 kcal) | 178.1±17.0 | 182.7±17.5 | <0.01 |
| Protein (g/1,000 kcal) | 33.3±6.2 | 32.9±6.6 | <0.001 |
| Fat (g/1,000 kcal) | 15.7±5.9 | 14.1±6.0 | 0.095 |
Table 2.
Values are presented as odds ratio (95% confidence interval), unless otherwise stated. P-values <0.05 were considered significant. Model 1 was adjusted for age, BMI, total energy intake, and macronutrient intake; model 2 was adjusted for age, BMI, smoking status, alcohol intake, physical activity, total energy intake, and macronutrient intake; and model 3 was adjusted for age, BMI, smoking status, alcohol intake, physical activity, total energy intake, macronutrient intake, household income, and education level.
BMI, body mass index; HDL, high-density lipoprotein.
Table 3.
Values are presented as odds ratio (95% confidence interval), unless otherwise stated. P-values <0.05 were considered significant. Model 1 was adjusted for age, BMI, total energy intake, and macronutrient intake; model 2 was adjusted for age, BMI, smoking status, alcohol intake, physical activity, total energy intake, and macronutrient intake; and model 3 was adjusted for age, BMI, smoking status, alcohol intake, physical activity, total energy intake, macronutrient intake, household income, and education level.
BMI, body mass index; HDL, high-density lipoprotein.
REFERENCES
- TOOLS