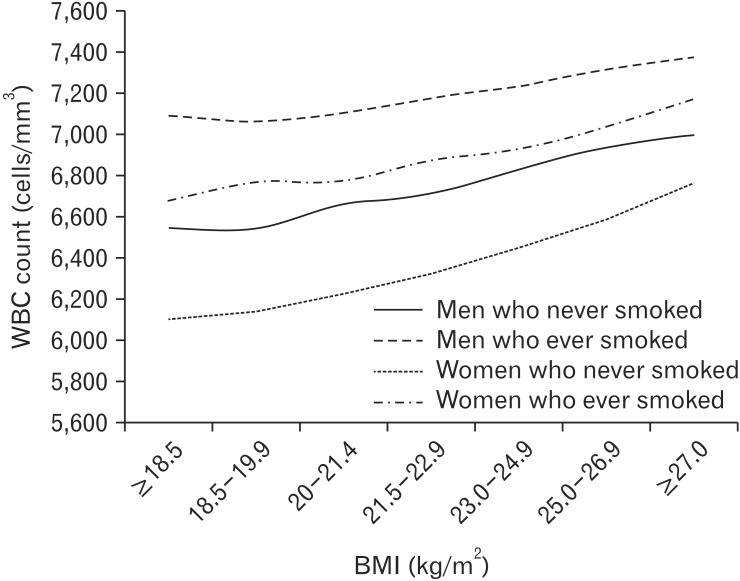
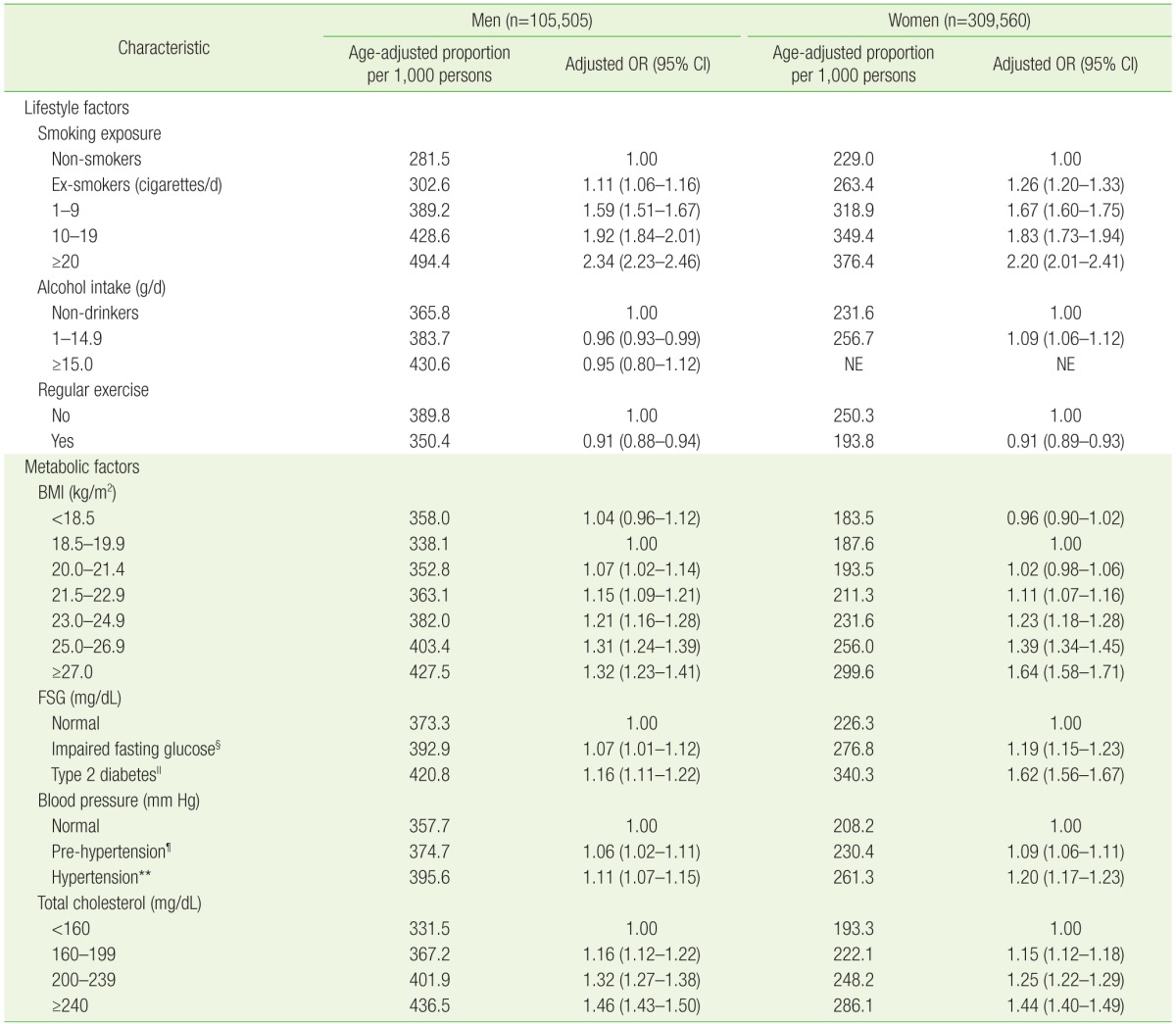
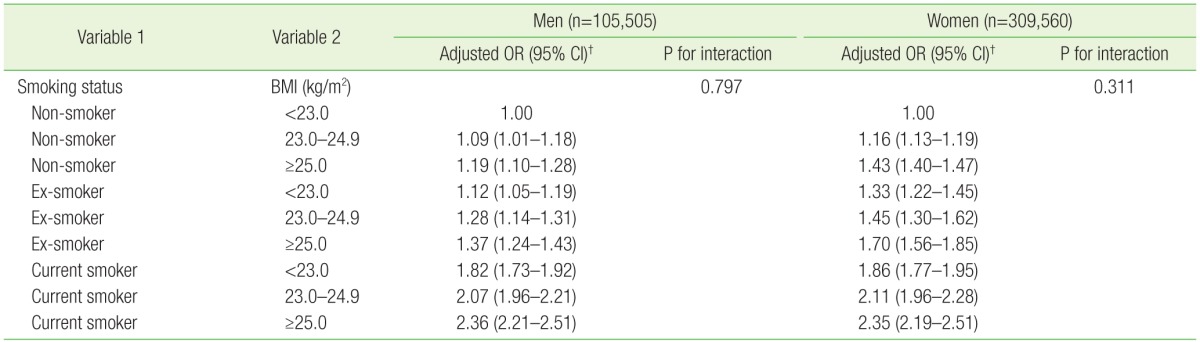
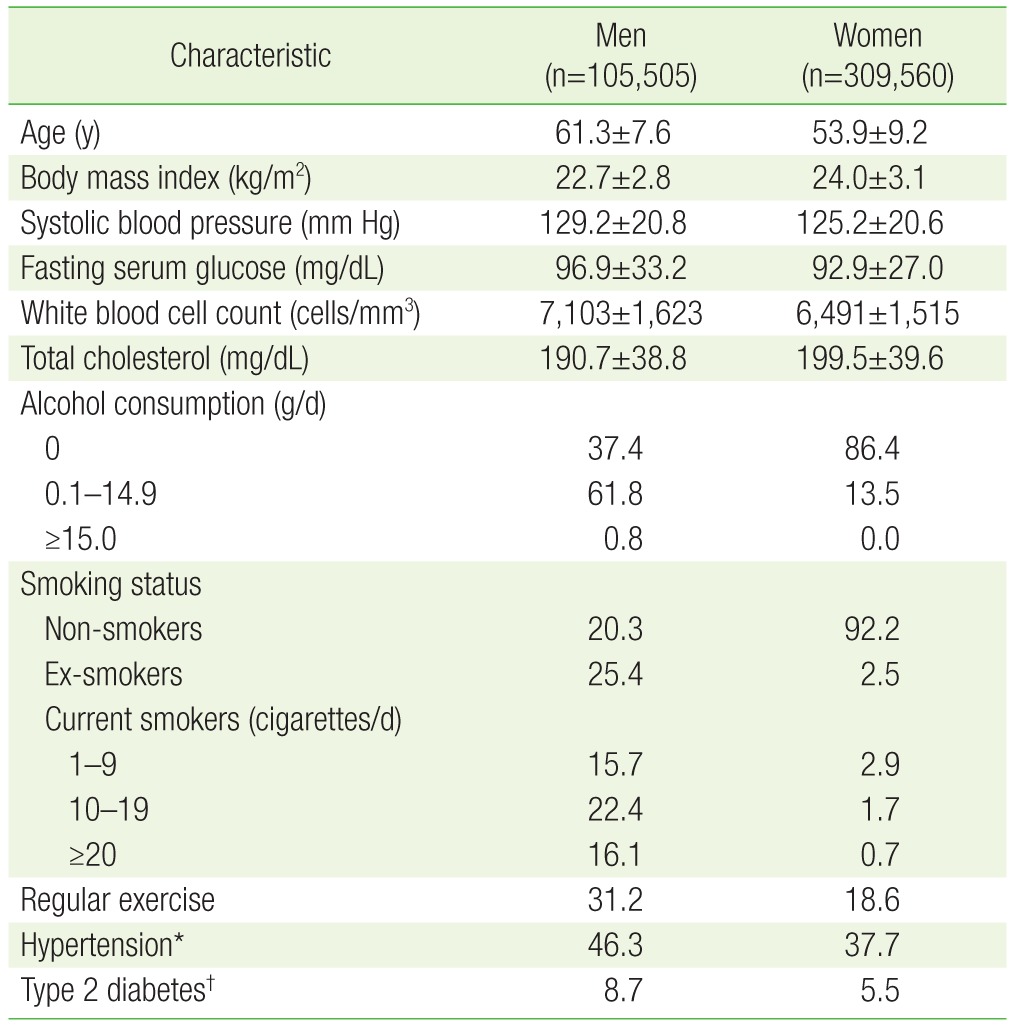
1. Ross R. Atherosclerosis: an inflammatory disease. N Engl J Med 1999;340:115-126. PMID:
9887164.


2. Libby P, Ridker PM, Maseri A. Inflammation and atherosclerosis. Circulation 2002;105:1135-1143. PMID:
11877368.


3. Corre F, Lellouch J, Schwartz D. Smoking and leucocyte-counts: results of an epidemiological survey. Lancet 1971;2:632-634. PMID:
4105947.


4. Petitti DB, Kipp H. The leukocyte count: associations with intensity of smoking and persistence of effect after quitting. Am J Epidemiol 1986;123:89-95. PMID:
3940445.



5. Nakanishi N, Yoshida H, Okamoto M, Matsuo Y, Suzuki K, Tatara K. Association of alcohol consumption with white blood cell count: a study of Japanese male office workers. J Intern Med 2003;253:367-374. PMID:
12603505.


6. Verdaet D, Dendale P, De Bacquer D, Delanghe J, Block P, De Backer G. Association between leisure time physical activity and markers of chronic inflammation related to coronary heart disease. Atherosclerosis 2004;176:303-310. PMID:
15380453.


7. Herishanu Y, Rogowski O, Polliack A, Marilus R. Leukocytosis in obese individuals: possible link in patients with unexplained persistent neutrophilia. Eur J Haematol 2006;76:516-520. PMID:
16696775.


8. Mugge A, Lopez JA. Do leukocytes have a role in hypertension? Hypertension 1991;17:331-333. PMID:
1999364.


9. Nakanishi N, Yoshida H, Matsuo Y, Suzuki K, Tatara K. White blood-cell count and the risk of impaired fasting glucose or type II diabetes in middle-aged Japanese men. Diabetologia 2002;45:42-48. PMID:
11845222.


10. Lohsoonthorn V, Dhanamun B, Williams MA. Prevalence of metabolic syndrome and its relationship to white blood cell count in a population of Thai men and women receiving routine health examinations. Am J Hypertens 2006;19:339-345. PMID:
16580566.



11. Ishizaka N, Ishizaka Y, Toda E, Nagai R, Yamakado M. Association between cigarette smoking, white blood cell count, and metabolic syndrome as defined by the Japanese criteria. Intern Med 2007;46:1167-1170. PMID:
17675764.


12. Kawada T. Smoking-induced leukocytosis can persist after cessation of smoking. Arch Med Res 2004;35:246-250. PMID:
15163468.


13. Martey CA, Pollock SJ, Turner CK, O'Reilly KM, Baglole CJ, Phipps RP, et al. Cigarette smoke induces cyclooxygenase-2 and microsomal prostaglandin E2 synthase in human lung fibroblasts: implications for lung inflammation and cancer. Am J Physiol Lung Cell Mol Physiol 2004;287:L981-L991. PMID:
15234907.


14. Pratley RE, Hagberg JM, Dengel DR, Rogus EM, Muller DC, Goldberg AP. Aerobic exercise training-induced reductions in abdominal fat and glucose-stimulated insulin responses in middle-aged and older men. J Am Geriatr Soc 2000;48:1055-1061. PMID:
10983904.


15. Liu S, Manson JE. Dietary carbohydrates, physical inactivity, obesity, and the ŌĆśmetabolic syndromeŌĆÖ as predictors of coronary heart disease. Curr Opin Lipidol 2001;12:395-404. PMID:
11507324.


16. Stewart KJ. Exercise training and the cardiovascular consequences of type 2 diabetes and hypertension: plausible mechanisms for improving cardiovascular health. JAMA 2002;288:1622-1631. PMID:
12350193.


17. Imperatore G, Cheng YJ, Williams DE, Fulton J, Gregg EW. Physical activity, cardiovascular fitness, and insulin sensitivity among U.S. adolescents: the National Health and Nutrition Examination Survey, 1999-2002. Diabetes Care 2006;29:1567-1572. PMID:
16801580.


18. Sharma AM. Adipose tissue: a mediator of cardiovascular risk. Int J Obes Relat Metab Disord 2002;26(Suppl 4):S5-S7. PMID:
12457291.



19. Friedman GD, Selby JV, Quesenberry CP Jr. The leukocyte count: a predictor of hypertension. J Clin Epidemiol 1990;43:907-911. PMID:
2213079.


20. Shankar A, Klein BE, Klein R. Relationship between white blood cell count and incident hypertension. Am J Hypertens 2004;17:233-239. PMID:
15001197.



21. Orakzai RH, Orakzai SH, Nasir K, Santos RD, Rana JS, Pimentel I, et al. Association of white blood cell count with systolic blood pressure within the normotensive range. J Hum Hypertens 2006;20:341-347. PMID:
16511508.



22. Gillum RF, Mussolino ME. White blood cell count and hypertension incidence: the NHANES I epidemiologic follow-up study. J Clin Epidemiol 1994;47:911-919. PMID:
7730895.


23. Elkind MS, Sciacca RR, Boden-Albala B, Tondella ML, Feikin DR, Fields BS, et al. Leukocyte count is associated with reduced endothelial reactivity. Atherosclerosis 2005;181:329-338. PMID:
16039287.


24. Rosenbaum M, Leibel RL, Hirsch J. Obesity. N Engl J Med 1997;337:396-407. PMID:
9241130.


25. Syvanne M, Taskinen MR. Lipids and lipoproteins as coronary risk factors in non-insulin-dependent diabetes mellitus. Lancet 1997;350(Suppl 1):SI20-SI23. PMID:
9250279.


26. Boden G. Effects of free fatty acids on gluconeogenesis and glycogenolysis. Life Sci 2003;72:977-988. PMID:
12495777.


27. Staehr P, Hother-Nielsen O, Landau BR, Chandramouli V, Holst JJ, Beck-Nielsen H. Effects of free fatty acids per se on glucose production, gluconeogenesis, and glycogenolysis. Diabetes 2003;52:260-267. PMID:
12540595.


28. Jee SH, Park JY, Kim HS, Lee TY, Samet JM. White blood cell count and risk for all-cause, cardiovascular, and cancer mortality in a cohort of Koreans. Am J Epidemiol 2005;162:1062-1069. PMID:
16221804.



29. Lee CD, Folsom AR, Nieto FJ, Chambless LE, Shahar E, Wolfe DA. White blood cell count and incidence of coronary heart disease and ischemic stroke and mortality from cardiovascular disease in African-American and White men and women: atherosclerosis risk in communities study. Am J Epidemiol 2001;154:758-764. PMID:
11590089.